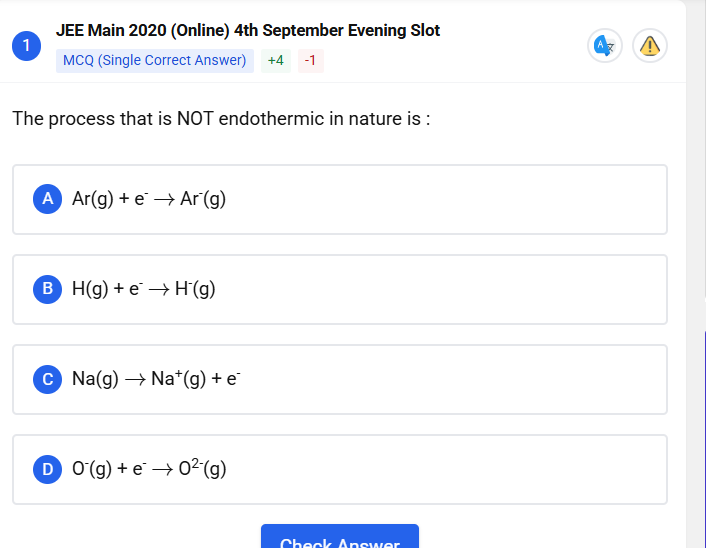
241 Replies
oh yes d bhi nahi hoga
@Dexter
Note for OP
+solved @user1 @user2... to close the thread when your doubt is solved. Mention the users who helped you solve the doubt. This will be added to their stats.adding electron to anion is alvays endothermic
so confused betveen b and c
C
rong
What?
galat
Ab energy to dekho lagegi electron nikaalna hai to
Daalna hai tab bhi lagegi 'cause hydride isn't really a stable species
Very reactive
ic
Ahmm weird
;-;
See I think D
D kyun nahi hogam
vould it be B because to remove an electron from sodium, you have to give energy?
Yeah
O2- na
Very prevalent
.
Octet bhi poora us bichare ka
Naah
Don't generalise like that
module me yahi diya hai ;-;
I mean.......
Net process exothermic hofa
i think in ncert too let me ceck
Ye sab sasti baate yaad mat karo
You always have to look at what species you adding electron to
Mai N+ ko electron dunga to that wouldn't be endothermic
The step mentioned here could be endothermic too
Oh wait aniob
Vahi 🙂
Yea sory
See O to O2- should be exothermic
O- to O2- vala step could be endothermic
vhy?
O is very electronegative
Can bear negative charge
oh right
itll give energy
to achieve stable
But negative pe or negative daalna is weird
Yeah
Please clear my doubt I know atom releases energy when it gets stable however this ques shatters that
morning
kya hua yaha
No charged species is more stable than a neutral species buddy:)
Chem se maut ka khela
ioc
Hmm ?
Real
Yeah exactly
are mai bemari me maut ka khela kar rhi aap yaha
okk okkkk
I will probably die a random day doing chem and maths
Mai chai pee aaun yaar canteen band ho jayegi 😭🙏
Aake dekhta hun ise
me when phy
If you think that way study from vinay uppal
Please don't bua ji
😭

,rotate
The person is not for you
bhyi

2 ans saath me
hn sahi to hai
replusion hoga
Huhh
That's...
Idk
@iTeachChem ?
Sir 😭🙏
kya jhol maal hai ye
ans kya diya hai?
Huh why ?
i dont feel like ve can assume this everyvhere because hou do u define "high e-e repulson"
hydrogen vaala optn
She's studying for neet and IAT
Not for jee advanced
Time to see a oneshot for periodic table revision
First gotta get her basics right
Vinay uppal apne jee mains ke one shot or full length course mai basics bhi karwate hai
b ok
Mai bhi dekh lun kya lmao
Hadn't reviewed this thing for a long while tabhi galti hui shayad
.oneshot
@Enamine is this correct (ignore the "vould it be B" part)
Tell this to a fella who is preparing for jee 6 months before jee
.padh
isme not endothermic bola to it is asking ki exo konsa hai
to exo is it gives out energy
ok?
pehle itna clear kar lete hai
yes
I'll get back to you
My brain is not braining rn
haan haan take ur time
to aage me ik ik karkr option dekhte kya hai
okay
Let's say when we add electron to that anion it gets stable. According to what I studied initially in chem bonding atoms releases energy after getting stable
1. argon hai
noble gas
would never give energy when taking up an electron
uska octet disturb hoga
ye hata do
fine till here?
yea
Haa
all ik is 1 nahi hoga
b last me karte ok?
oka
Oh hell naw it seems a guy turned on camera in class while in baniyaan 😭
c me it is sodium to sodium ion with electron
ab ye chiz hum kab possible kar payenge jab hum usko energy denge
kyunki even though uska hai octet after loosing isme itne paas hai electron ki energy need ho jata hai
iski na uhm mai apko energies bhi deti hu kiitni lagri sodium me
ew disgusting man
💀
so basically idhar IE ka khel hai
d me we have O- to O2- which is ki hume usme ik electron aur add karna hai
jaha usme alredy ik electron hai
ye kya ho gya second electron gain enthaply
to ye to obv nhi hoga exothermic
yes exactly
now for the last one
which is b
oka
ye bhi samjha
ye samjha do h ka kyu endo h
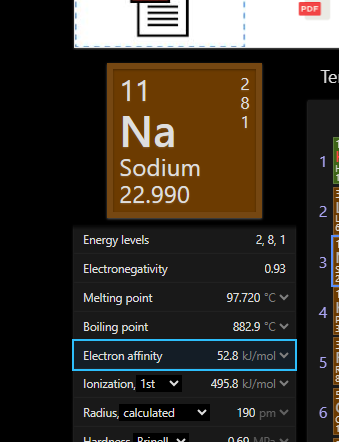
kam hai kafii par isme bhi dena hoga energy
How is the affinity positive?
Should negative no?
it is negative
according to
my book
Yeah should be
haan
only Be and N and noble gases have +ve ie right
Sir saying he looks smart
vhy are u on dc during class 😠
.padh
Although bro got chieseld jawline
Cause it's a revision class ? I joined only for attendence
oh
fair enuf
tbh ye exothermic nhi hai
._.
Bua ji periodic table padha do tbh
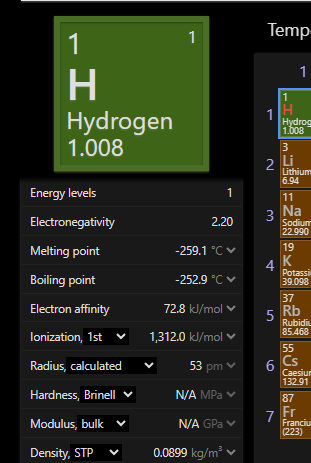
maine kabse revision nhi kiya hai 😭 adha foggy hai
organic bolo hn thik hai par ye waale nhi yaad
isme kya dekhu
hydrogen me jo effect sodium me vahi chiz more amount me hai
IE value 😭
h
oh
mb
h2o adding h and oh
handekhi
omg mai pgl ho gyi hu
😭
._.
peak ioc
to isme b kyu hai i dont get it
💀
i think i get it
nvm i dont
One more thing to add this is the class of SRG batch
omg
even more
😭
thingg is all 4 of these are endothermic process so idk which idiot made this question
lke the values are in 3000 for oxygen
j
around 400 ish for na
oxygen ke liye toh ye diya hai
the other 2 u saw
peak nta
BROO nta
i think you vere supposed to get confused at option 2 during the exam, say "baadme dekhenge", do the other options, then by process of elimination mark 2
what is wrong
but the ans is none
I think we need to summon sir
varun ji already did
even the first one needs energy
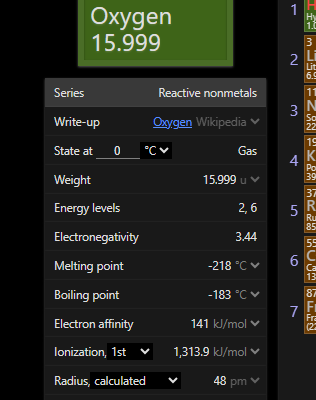
atleast a vill be endothermic right
and also c
Naaah
First one is exothermic
so the confusion is betveen b and d!!!
statistically none is but ok
😭 this is from ptable.com
i am not saying on my own
Really no?
It's not accounting for signs
Values ka idk
It's not accounting for the signs
yes
none is
it is
that is the worst part
Check pubchem or something
That's more reliable as well ngl
I'll read up on this though
Batata hun fir
hn mai karti
@Jeepaglu
You move ahead
I'll see this one and ping you
alright tysm
says the same thing
Sign?
isme sign nhi hai
Ye websites bhi na……
Ionisation energy to positive hi hogi thik hain
Electron affinity ka karna
meri physical heath aachi nhi hai chem too dur ki baat 😭
zumdahl ya atkins check karu?
Abeyy yaar take care
I will don’t worry
Will ping you guys in a bit
ok okk thnkss
same thoda merko bhi jana hai
Hmm hmm kar dunga
Will ping both you and jeepaglu
ek
baar
ek baar
ye bhi samjha dena
do baar
Dekho
O se O- jaana is feasible
Kyun?
O is electronegative
haan
But ab already negatively charged chiz pe or negative charge daalna
That’s highly unfavourable until you have something around
Like counter ion and all
makes sense
Kisi chiz se react karke basically vo energy release nahi karwa sakta taaki overall exothermic ho jaaye
Yahi chiz agar solution me hoti to O se O2- would be exothermic
Gas phase me hai isiliye endothermic hai
ye vaala part ek baar aur
Ab Mai tumhara original question khule dimag se dekhta hun
Like socho agar O- se O2- me energy lagti hai bohot jyada
Agar solvation energy ke naam pe next step me bohot saari energy release ho jaaye
To overall process exothermic ho jayega na?
solvation energy?
H2O me agar koi ion hota hai
To water ke molecules use gher lete hain na
Ya kisi bhi solvent me hota hai basically
Agar polar solvent hai to ions gher lenge na use
That electrostatic potential energy generated due to that charge separation
haan
That’s very negative
(Kitna negative depends upon the ion)
ic
makes sense
.
all the best 🙏
@Sephrina your glasses are of negative power or positive ?
negative
Accha you can relate then. Do you keep a printed periodic table on wall ? If yes are you able to see the electron config written on it clearly ? 😭
I can't
nah i cant see it 😭
I have spent an hour finding a perfect periodic table pdf which I can print on A4 paper
And still see electron config written on it
:kekw:
us
So what did you do in end ?
If I don't find a solution in a week
I will just go and get laser surgery
i just keep one website open
the ptable wnt ever be visible like that to me
I don't want to use my laptop everytime so its hectic for me
same par option nhi hai
That's something you did by elimination
How do you say the H to hydride thing is exothermic
I mean we aren't technically talking about bond breaking and forming
Aise naked atom ko electrons dena and lena
Isme usually energy is required
For Oxygen to oxygen (1-)
Being exothermic is understandable
Na to Na+ being endo is understandable
('cause it's in gas phase; no solvation)
Argon vala option is also understandable as you explained
But hydride vala thing......
I'm not really getting how is that exothermic
vait, o- + 1e = o-- is an exception for being endothermic?
this
isnt it all anions + 1e = endothermic
,
hn lekin oxygen ka vaha octet ho jata haina
Are you're giving it to an electronegative element
🤡 NTA
ok sir makes sense
But yahan to it's easy explanation isn't it 😭
Why mug here
As far as I can see, there are already two lone pairs
2 more electrons
Basically what I wanted to say was too much interelectronic repulsions
Considering it's a second period atom
dont get me wrong i love chemistry but sometimes the reason for my fried brain is chemistry
Case in point
stop studying chemistry lord
That's something I told him above
Just 'cause this is in gas phase
😔
So it seems like too much of energy is required
keep studying chemistry
But when you have a counter ion
You have a solvent
Then it compensates for it
yea its pretty interesting
its like that one game vhere you say 'this game sucks' then continue playing it
cuz the game itself is fun
that ONE thing is frustrating
See it's all logical
Just that time lagta hai
That solvation and lattice thing chemical bonding me aa jaati hai generally
If not there to 12th me solutions, electro somewhere at least
ic
so for rn, just solve using elim?
and ncert
yes
iteachchem
Transcription requested by Abhi
या ब्रो, MCQ है और क्या करना है, वही तो करना है.
iteachchem
Transcription requested by Abhi
देखो अगर bond form हो रहा है तो exothermic है, ठीक है, so A, B, or D में से you have to pick, C में you are breaking a bond technically, so that is obviously endothermic, A तो argon है, it does not want an electron whatever you do, so you got to give it a lot of energy to accept that, Oxygen का भी second वाला endothermic step होता है, that's an exception, मग कर लो NCRT में है, must be B,
iteachchem
Transcription requested by Abhi
And also, if you want to know PQ, I told you, everything that forms bonds must be exothermic.
iteachchem
Transcription requested by Abhi
So, elimination says like you give all of those are exceptions to this idea. He you know, forming bondage is atomic.
iteachchem
Transcription requested by Abhi
अरे NCRT पढ़ ले भाई.
Yeah that's something that also could do the same
Like enhance the formation of it
iteachchem
Transcription requested by Abhi
इतना सोचो मत भाई, नेचर में understand करना मुश्किल है, I have told you a simple idea, bond forming gives off energy, bond breaking requires energy, think of this like a bond, that's what it is right, see what is a bond, दो electron, electron का यहां से वहाँ जाना या share होना, in this case, one half of that is happening,
iteachchem
Transcription requested by Abhi
Can you please read NCRD?
iteachchem
Transcription requested by Abhi
This is one of those things that you just have to mug. If you want to know why, you're going to have to read a lot of books.
iteachchem
Transcription requested by Abhi
But this is also another thing that's tested a lot in JEE and NEET, because mug vomit works in all these exams, like that thing, what is it called? Inert pair effect, zabarasti puchenge bhai toh mug karo.
iteachchem
Transcription requested by Abhi
Yeah, the second step is always endothermic.
iteachchem
Transcription requested by Abhi
But oxygen case mein bahut zyada endothermic hai because of other
**** , forget it like I don't want to get into it.ABHI BRO
Abhi
😭
Stop
Or I'm gonna ban you
stop
iteachchem
Transcription requested by Abhi
What you are saying is right but the confusion arises कि अगर endothermic है then why does the O2- ion get formed in the first place वो ये बच्चे को मैं यहां बोलेंगे तो he will get confused and stop studying chemistry But we will learn it by 12th which is the concept of lattice energy because that stabilizes the compound Simple terms right, that's it
..
iteachchem
Transcription requested by Abhi
No, you don't need a solvent, no. You have solid compounds that get formed, so lattice energy is the main thing.
iteachchem
Transcription requested by Abhi
But let's stay on point, एक चीज में एक चीज का answer देते हैं, वो हमने दे दे और इसको बन चाहते हैं, let's not spend anymore time on this.
yes sir that makes sense
thanks alot everyone
iteachchem
Transcription requested by Abhi
लॉजिक भी तो बताया, एलेमनेशन है और हाइड्राइट तो सिंपल है, all things where bonds form are exothermic and exceptions, all of the other three things are exceptions to the rule and generally second affinity, electron affinity will always be endothermic, वो उसने बताया क्यों?
Done.
ill close this nou
He's gone
Just saw the msgs
Sorry sir
I was too busy reading the transcripts
Agli baar ping kar dijiye ga sir instant pta chal jaayega mereko
+solved @iTeachChem @Sephrina @Enamine
Post locked and archived successfully!
Archived by
<@1386389961196048405> (1386389961196048405)
Time
<t:1751378930:R>
Solved by
<@1035556259417571408> (1035556259417571408), <@888280831863451688> (888280831863451688), <@984016629119713290> (984016629119713290)