Reimer tiemann mech
Is wle step me oxygen ne attack kyu nahi kia? Pi bond se kyu karwana pada
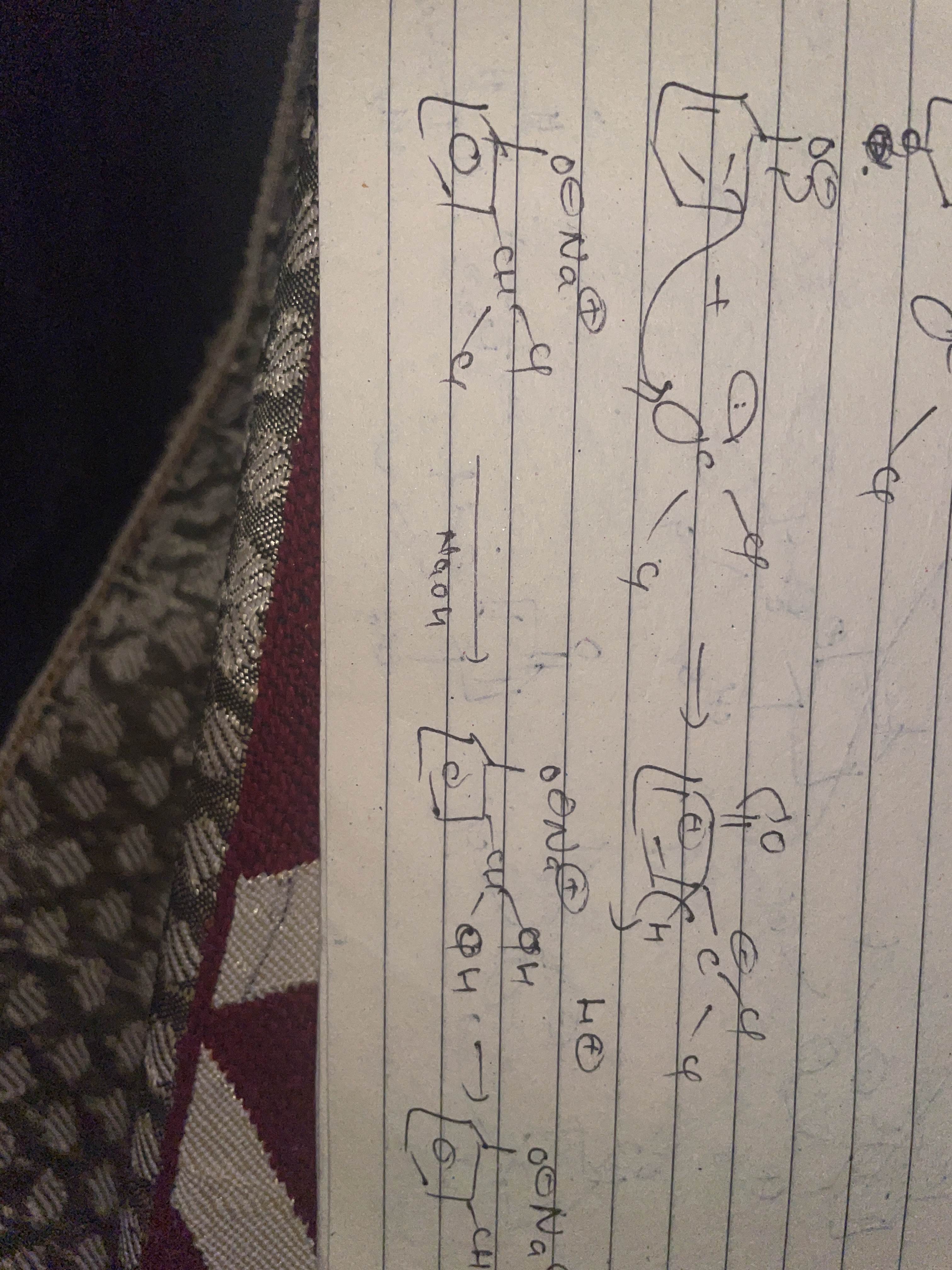
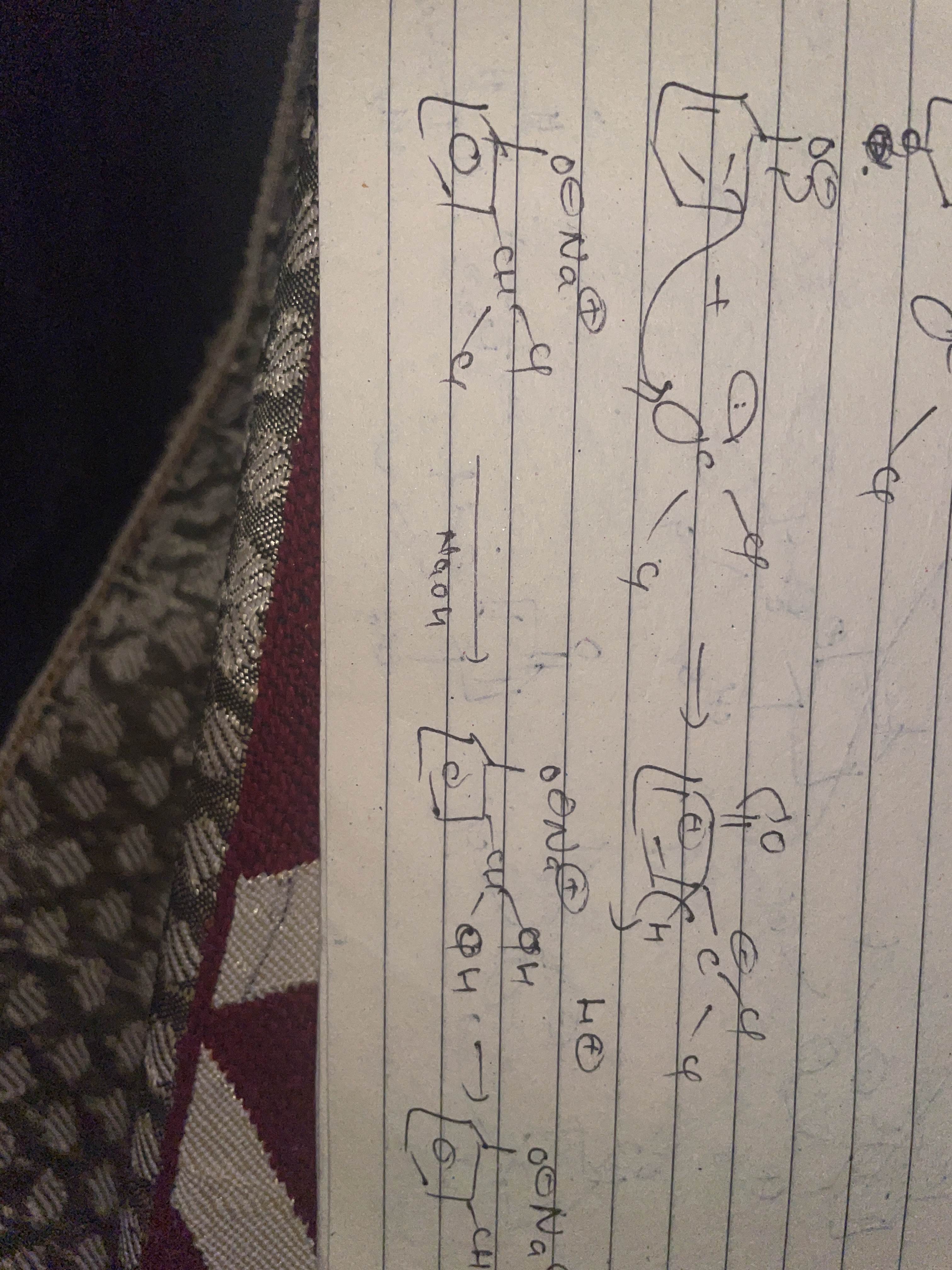
Join the Discord to ask follow-up questions and connect with the community
A Study Community for Everyone and Everything. We have weekly sessions on Spirituality, Math and Science! JEE, NEET prep? All doubts are crowdsourced. Hare Krishna!
1,725 Members
Join